Mathematical Uncertainty Principles
How to understand a core idea from Quantum Mechanics.

I’d like to give a fairly simple account of why uncertainty principles exist in quantum mechanics (and math). I often see in movies and sci-fi books, not to mention real-life discussions, a misunderstanding about what they mean.
Recall the classic Heisenberg form in quantum mechanics. It says:
We cannot know the exact momentum and position of a particle simultaneously.
Preliminary Interpretations
First, I like the above phrasing a bit better than a common alternative: we cannot measure the momentum and position simultaneously.
When I took quantum mechanics as an undergrad, I thought I understood this. There was a clear picture in my head. When you measure the position, you bump the particle and hence lose track of the momentum and vice versa.
Although I guess this is technically true, it gives the wrong idea. This makes it sound like we don’t have good enough measuring equipment. Maybe in a hundred years, our tools will get better, and we will be able to make more precise measurements to do both at once.
This is wrong and completely misunderstands the principle.
When I got to grad school, I realized that “uncertainty principles” exist all over the place in a purely mathematical setting. The Heisenberg uncertainty principle is actually a purely mathematical fact that comes from the momentum and position operators.
This means that from a theoretical perspective, we can never “know” both simultaneously even mathematically without using measuring devices.
I don’t want to go too deep on what it means to “know” something. It’s a bit off-topic for the main point of this article.
In some sense, the uncertainty principle should say that it makes no sense to ask for the momentum and position of a particle (although this again is misleading because we know the precise amount of uncertainty in attempting to do this).
It’s like asking: Is blue hard or soft? It doesn’t make sense to ask for the hardness property of a color. You cannot even write down an equation (a wavefunction for a particle) that has a precise momentum and position at the same time.
The Math Behind It
Here’s the mathematical formalism that lets this fall out easily. To each observable quantity (for example: momentum and position) there exists a Hermitian operator.
If you haven’t seen this before, then don’t worry. The only fact we need about this is that “knowing” or “measuring” or “being in” a certain observable state corresponds to the wavefunction of the particle being an eigenfunction for this operator (definition will be given).
Suppose we have two operators A and B corresponding to observable quantities a and b, and it makes sense to say that for Ψ we can simultaneously measure properties a and b.
This means there are two number λ₁ and λ₂ such that
AΨ = λ₁ Ψ and BΨ = λ₂ Ψ.
That is the definition of being an eigenfunction.
The commutator of two operators is just doing AB and then subtracting doing BA.
If we apply the commutator to Ψ, look at what happens:
[A,B]Ψ = ABΨ — BAΨ
= A λ₂ Ψ — B λ₁ Ψ
= λ₂ λ₁ Ψ — λ₁ λ₂ Ψ
= 0.
It’s just the definition and then applying the definition of being an eigenfunction. There’s nothing advanced or tricky going on in the simplifications.
Mathematically speaking, a particle that is in a state for which it makes sense to talk about having two definite observable quantities attached must be described by a wavefunction in the kernel of the commutator.
Don’t worry about the terminology there. It’s just a basic fact about operators that if you can know both simultaneously for a given function (or distribution), then the commutator applied to that function gives you 0.
Therefore, it never even makes sense to ask for both if the commutator has no (non-trivial) kernel.
Now for the Heisenberg uncertainty principle in quantum mechanics, all we must do is compute the commutator of the momentum and position operator and see that it has no kernel (except for the 0 function which doesn’t correspond to a legitimate wavefunction).
You could check Wikipedia or something, but the position operator is given by xf= xf and the momentum is given by pf=-i ħ (d/dx)f.
Now we compute:
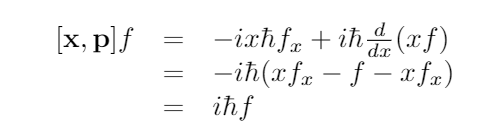
This shows that the commutator is a constant times the identity operator. It can never be 0!
Therefore, it makes no sense to ask for a definite position and momentum of a particle simultaneously. There isn’t even some crazy, abstract purely theoretical construction of a particle that can have that property.
It’s worth pointing this fact out because there are operators for which you often can’t know two properties simultaneously but for which there are special particles where you can.
This also shows that we can have all sorts of other mathematical uncertainty principles by checking other operators. A famous one is the Fourier transform in which there is a fundamental uncertainty in knowing both the frequencies and its localization in time.
Takeaways
The Heisenberg Uncertainty Principle doesn’t have to do with quantum weirdness or the observer collapsing the wavefunction. It doesn’t have to do with imprecise measuring equipment or changing the particle by measuring it.
Uncertainty principles exist all over mathematics, and as the above shows, it is a purely mathematical phenomenon of operators. To me, this is one of the better ways to interpret it.